The emergence of new strains of PRRS virus together with limitations in antibiotic use are shifting paradigms in swine production. Throughout Spain, it is a fact that pig mortality from weaning to market has increased by 4% due to an increase in health problems. Every day we learn about new tools to monitor many of the pathologies that affect our farms, but we must take a different approach to the usual problems and this is where the work of the production veterinarian lies: in knowing how to change. To do this, we have to rely on new research (books, scientific articles, etc.) as well as on the animals themselves.
It is now well documented that the intestinal microbiota clearly influences the modulation of inflammatory responses and, therefore, any digestive disorder that leads to changes in the microbiota possibly causes a reduction in alveolar macrophages and a change in the Th1 and Th2 (T Helper Leukocytes) immune response. As a consequence, there may be an increase in the inflammatory response at the pulmonary level and also an increase in the recurrence and persistence of pulmonary diseases.

Clinical case
We are dealing with a farm of about 2,000 breeding sows located in the Spanish region of Murcia. The property is approximately 100 hectares, so the different production phases are located far from one another. The breeding sows are in one site, the nurseries are located 1 km away, and the finishing barns are distributed about 3 km from the breeding sows.
The breeding sow farm is PRRSv positive and is currently stable. It has a monthly monitoring plan that includes collecting tip tongues from stillborn piglets, differentiating between piglets from multiparous and primiparous sows. This monitoring consists of PCR testing for PRRS and PCV-2, with the objective of controlling both viruses on the farm. During this period, the stillborn tip tongue analyses were negative for PRRS and PCV-2. When a sample is positive for PRRS virus, we perform sequencing by ORF5 to check if it is a new strain coming from a lateral introduction of the virus which would indicate failures in external biosecurity.
The farm is positive for Brachyspira hyodysenteriae but, through the use of autogenous vaccines, the clinical signs are controlled and no symptoms are observed in the sows in production or in the replacement gilts, which have the said autogenous vaccine included in their vaccination program. The clinical disease is considered controlled, although occasionally symptoms compatible with swine dysentery appear in the finishing barns when the pigs weigh approximately 50-80 kg, coinciding with the loss of the immunity that had been acquired through the dams.
A certain level of tail biting is observed in finishing, which we consider an indicator of the presence of dysentery even in cases where we do not observe a clearly compatible clinical picture. If the pigs have subclinical disease and, therefore, inflammation in the colon, they tend to be more nervous and move their tails insistently. As we know, this is like a magnet for their pen mates who will start biting each other. Once the abnormal behavior is established it is difficult to control, as chronic inflammation induces anxiety-like behavior.

Photo 1. Pigs with tail biting.
Start of the problem
The animals are moved to finishing barns with 6,000 places that are filled for four consecutive weeks. One month after animal entry, a respiratory clinical signs begin with severe dyspnea and anorexia. The decision is made to administer an antibiotic treatment with soluble florfenicol in the drinking water (combined with an anti-inflammatory on the first day to increase water consumption), but after this treatment, deaths continue and there is no improvement in the animals. Lungs from dead animals with large areas of pulmonary consolidation are sent to the laboratory. The decision was made to administer an injectable antibiotic treatment (Marbofloxacin) to all the pigs, The idea was, on the one hand, to ensure that each animal received the appropriate dose and, on the other hand, to reach the lungs with an effective concentration of antibiotic. In spite of an improvement in the animals' overall condition, after two weeks a relapse is observed and we are still unable to control the losses that occur. The clinical picture returns with the same intensity and continues to cause many deaths (Photos 2 and 3).

Photo 2. Pig with cyanosis.

Photo 3. Pigs in the barn with fever.
Lung samples were sent to the laboratory from the dead animals and PCR was requested for PRRS, PCV-2, and Influenza A. All the samples were negative for PCV-2 and Influenza A and positive for PRRS virus, so we proceeded to sequence the virus by ORF5 to see if a high virulence strain had entered the farm that could explain the severity of the clinical symptoms.
Table 1. Analysis of lung and oral fluid samples that were taken in the finishing barn.
| Date | Finishing barn | Sample | PRRSv | PCV-2 | Influenza A |
|---|---|---|---|---|---|
| Nov-22 | A | Lung | PRRSv (Ct 28) | - | - |
| Nov-22 | B | Lung | PRSSv (Ct 20) | - | - |
| Nov-22 | C | Oral fluids | PRRSv (Ct 30) | - | - |
| Nov-22 | D | Oral fluids | PRRSv (Ct 36) | - | - |
However, a significant concern came up: normally, a newly introduced pathogen, if it has a low prevalence, produces clinical symptoms at the end of the finishing period and, as its prevalence increases, it does so in younger and younger animals. However, this is not what happened in this production pyramid, so we thought that antibiotic treatments would reduce the impact of the disease, but this did not occur.
When the PRRS virus sequencing was obtained, it was found to be the same strain since there was a phylogenetic homology with the production system resident strain. However, it had never behaved this way in the finishing phase. Previously, respiratory symptomatology appeared, but it was easily controlled with antibiotic treatment (florfenicol).
What was happening with the finishing pigs to explain this abnormal pattern?
At this time a new way of approaching the infection is being proposed, since there is an ever-increasing knowledge of the gut-lung axis, and it is recognized that what happens in the intestine affects the severity of respiratory disease.
We wondered if there was any other pathology that was not pulmonary but rather intestinal and we isolated Brachyspira hyodesenteriae in the feces. This finding was not new, but we thought that it was controlled since the classic symptoms of mucohemorrhagic diarrhea were not present, but just some soft, loose feces that did not seem to have much clinical importance.
Could the presence of Brachyspira hyodesenteriae be one of the causes of the severity and persistence of clinical symptoms?
HYPOTHESIS: "The presence of Brachyspira hyodesenteriae and its impact on the intestinal microbiota could be affecting the modulation of inflammatory responses with an increase in the inflammatory response at the pulmonary level,also producing a reduction of alveolar macrophages and therefore creating an immune environment in which it was not possible to eliminate the PRRS virus with sufficient efficacy and also leading to an increase in recurrences and persistence of pulmonary diseases."
We proceeded to verify that the isolated strain of Brachyspira hyodesenteriae was still the farm`s resident strain and to determine the MICs for the different antibiotics that we had to use for its control.
Table 2. MIC of the different antibiotics for the isolated Brachyspira hyodesenteriae strain.
| Strain SV22-3448 | |||||
|---|---|---|---|---|---|
| Antibiotic range tested (μg/ml) |
MIC (μg/ml) |
Sensitivity interpretation | Range (μg/ml) | ||
| Tiamulin (0.063 - 8) | 0.5 | Intermediate |
≤ 0.25 > 0.25 - ≤ 2 > 2 |
Sensitive Intermediate Resistant |
La et al., 2016 |
| Valnemulin (0.031 - 4) | < 0.031 | Sensitive |
≤ 0.25 > 0.25 - ≤ 2 > 2 |
Sensitive Intermediate Resistant |
La et al., 2016 |
| Tylvalosin (0.25 - 32) | 8 | Intermediate |
≤ 0.25 - < 1.5 > 1.5 - ≤ 36 > 36 |
Sensitive Intermediate Resistant |
Pringle et al., 2012 |
| Lincomycin (0.5 - 64) | 32 | Intermediate |
≤ 4 > 4 - ≤ 36 > 36 |
Sensitive Intermediate Resistant |
Kirchgässner et al., 2016 |
| Tylosin (2 - 128) | > 128 | Resistant |
≤ 1 > 1 - ≤ 4 > 4 |
Sensitive Intermediate Resistant |
La et al., 2016 |
As we can see in Table 2, tiamulin appeared to be the best option, but at the farm level, considering the physical characteristics of the water in the Murcia region (poor quality and high salinity), this antibiotic does not provide good efficacy, making lincomycin the active ingredient that offers greater coverage for a better resolution of the clinical problem. We proceeded to establish an antibiotic treatment focused primarily on the control of swine dysentery at the subclinical level.
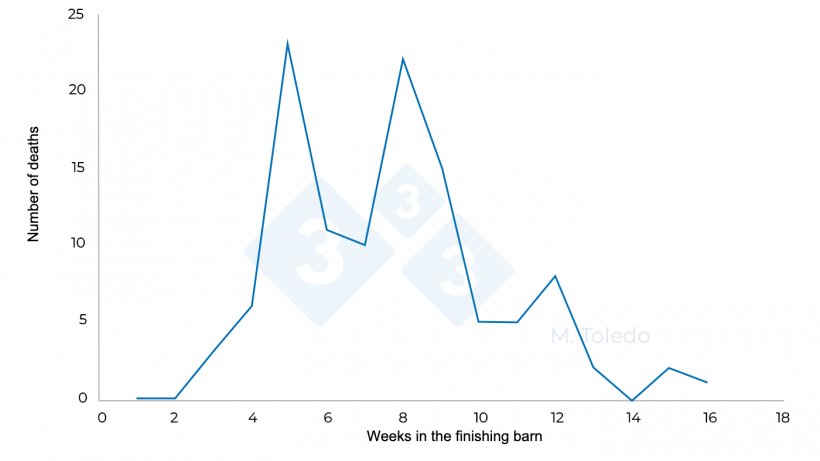
Figure 3. Evolution of the deaths in a finishing barn after medication: S4 - florfenicol in drinking water, S6 - injectable marbofloxacin, S8 -Tiamulin or Lincomycin in drinking water.
Conclusion
The successful approach to this clinical case was to control dysentery, even if we found it subclinically. In this way, the respiratory disease evolved in a much less aggressive way and without recurrences. Controlling dysentery also led to a reduction in the occurrence of new episodes of tail biting.
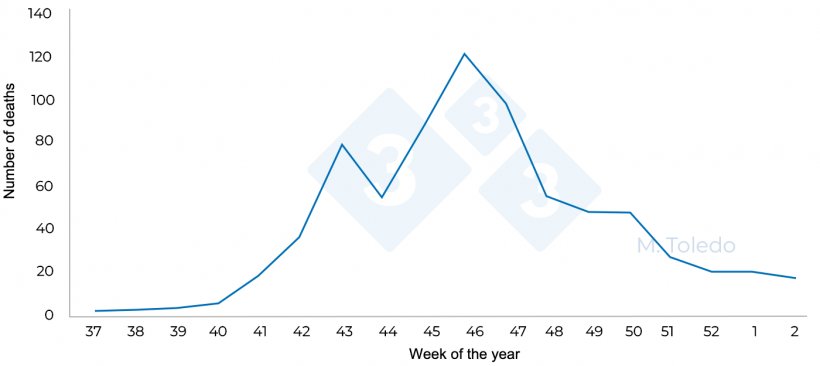
Figure 4. Total pig deaths at finishing in relation to the week of the year.
We must begin to approach respiratory diseases differently (especially the one causing us the most problems, PRRS virus), by monitoring and controlling enteric disorders that appear due to interactions between the different intestinal pathogens that are capable of producing dysbiosis. It is important not to consider these digestive pathogens individually as there are many interactions between them (Brachyspira spp, Lawsonia intracellularis, Escherichia coli, etc.). This alteration of the intestinal system may be modulating the intensity and duration of respiratory diseases and, therefore, complicating their control.